Ch 19.3 #1
A constant-volume
gas thermometer is calibrated in dry ice (that is, carbon dioxide in the solid
state, which has a temperature of –80.0°C) and in boiling ethyl alcohol
(78.0°C). The two pressures are 0.900 atm and 1.635 atm.
(a) What Celsius
value of absolute zero does the calibration yield?
What is the
pressure at (b) the freezing point of water and (c) the boiling point of water?
|
PV = nRT (V is constant) y = mx + b; Plot T vs P So find slope, then y-intercept m = (78 - -80) /
(1.635 - 0.9) m =
214.966 °C/atm Obtain y-int y =
m x + b 78 =
214.966(1.635) + b b =
-273.47 °C |
(a) y = mx + b y = 214.966(0) +
-273.47 0 K =
-273.47 °C (b) y = 214.966(x) +
-273.47 0 = 214.966(x) +
-273.47 x =
1.27 atm |
(c) y = m x + b T =
214.966 P + -273.47 y = 214.966(x) + -273.47 100 = 214.966(x)
+ -273.47 x =
1.74 atm |
Ch 19.4 #13
The active element
of a certain laser is made of a glass rod 30.0 cm long by
1.50
cm in diameter. If the
temperature of the rod increases by 65.0°C, what is the increase in (a) its
length, (b) its diameter, and (c) its volume?
Assume that the average coefficient of linear expansion of the glass is
9.00 ´ 10–6(°C)–1.
|
(a) ΔL = α L
ΔT ΔL = 9´10–6 (300 mm)65 ΔL
= 0.176 mm |
(b) area is
πr2…but diameter is just 2πr so still linear ΔL = α L ΔT ΔL = 9´10–6 (15 mm)65 ΔL
= 0.00878 mm |
(c) Volume is
Lπr2 (cubic)…so 3 α ΔV = 3α V ΔT ΔV = 3*9´10–6 (Lπr2) 65 ΔV = 3*9´10–6 (30π(1.5/2)2)
65 ΔV
= 0.0930 mm3 |
Ch 19.4 #15
A brass ring of
diameter 10.00 cm at 20.0°C is heated and slipped over an aluminum rod of
diameter 10.01 cm at 20.0°C. Assuming the average coefficients of linear
expansion are constant, (a) to what temperature must this combination be cooled
to separate them? Is this attainable? (b) What If? What if the aluminum rod were 10.02 cm in
diameter?
Why this equation?…we know at a certain temp their total length after
expansion (negative) will be equal to each other.
|
LAl + DLAl = LBr + DLBr LAl + LAl aAl DT = LBr + LBr aBr DT Solve for DT DT = (LAl - LBr) / (LBr aBr - LAl aAl) DT = (10.01 -
10)/(10*19x10-6 – 10.01*24x10-6) DT = -199 C° 20°C +
-199 C° > -273.15 °C; yes…it can be done |
b) DT = (LAl - LBr) / (LBr aBr - LAl aAl) DT =
(10.02-10)/(10*19x10-6 –10.02*24x10-6) DT = -396 C° 20°C +
-396 C° < -273.15 °C; no, we can’t go below absolute zero |
Alpha varies
slightly depending on source…lab manual aAl = 25x10-6 lecture book aAl = 24x10-6
Also…Brass is
an alloy…so alpha will vary also…
|
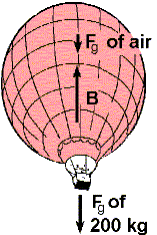
Ch 19.5 #33 The mass of a
hot-air balloon and its cargo (not including the air inside) is 200 kg. The
air outside is at 10.0°C and 101 kPa. The volume of
the balloon is 400 m3. To what temperature must the air in the
balloon be heated before the balloon will lift off? (Air density
at 10.0°C is 1.25 kg/m3.) Hint: Since n/V
is a measure of density we have n/V = (P/R) 1/T Pressure is constant since open
hole…so n/V is inversely proportional to Temperature |
|
|
|
mballoong – FB = 0 mballoong - (Vρairg
-Vρhot airg) = 0 mballoon
= Vρair - Vρhot air mballoon/V
= ρair -
ρhot air ρhot air = 1.25 – (200/400) ρhot air = 0.75 kg/m3 |
ρhot air/ρair = T/Thot 0.75/1.25 =
283.15/Thot Thot = 472 K |
|
Ch 19.5 #57
Consider an object
with any one of the shapes displayed in Table 10.2.
What is the percentage increase in the moment of inertia of the object when it
is heated from 0°C to 100°C if it is composed of (a) copper or (b)
aluminum? Assume that the average linear
expansion coefficients shown in Table 19.1 do not vary between 0°C and 100°C.
|
I = ∫r2dm Δr = ri
α ΔT rf – ri = ri α ΔT rf = ri + ri α ΔT rf = ri (1 + α ΔT) If /
Ii = ∫rf2dm
/ ∫ri2dm If /
Ii = ∫ri2(1 + αΔT)2dm /∫ri2dm If /
Ii = ∫ri2dm(1 + αΔT)2/∫ri2dm |
If /
Ii = ∫ri2dm(1 + αΔT)2/∫ri2dm If /
Ii = (1 + α ΔT)2 If /
Ii = 12 + 2 α
ΔT + α2ΔT2 Since α2ΔT2
<< 1, it cancels out If /
Ii = 1 + 2 α
ΔT If /
Ii - 1 = 2 α ΔT (If -
Ii) / Ii = 2 α ΔT |
|
(a) αCu =
17.0 x 10-6 (If -
Ii) / Ii = 2(17.0x10-6)100 (If -
Ii) / Ii = 0.340 % |
(b) αAl =
24.0 x 10-6 (If -
Ii) / Ii = 2(24.0x10-6)100 (If -
Ii) / Ii = 0.480 % |