#2, solar, 22, 27,
33, gasses, 51
|
Ch
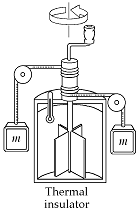
20.1 #2 Consider Joule's
apparatus. The mass of each of the two
blocks is 1.50 kg, and the insulated tank is filled with 200 g of water. What is the increase in the temperature of
the water after the blocks fall through a distance of 3.00 m? ans 0.105 °C |
|
|
|
We know Q = 0 in
Joules apparatus (insulated) ΔEint = Q + W; ΔEint = W Work = 2 blocks
(mgh) Work =
2(1.5*9.8*3) |
Work =
88.2 J ΔEint = 88.2 J = mcDT 88.2 J =
0.2*4186*DT DT = 0.105 C° |
|
Ch
20.2 #solar
We are going to
heat the water in a calorimeter cup using solar power. Our solar collector has an area of 10.00 m2,
and the intensity delivered by sunlight is 600 W/m2, how long
does it take to increase the temperature of 300 cc of water from 20.0°C to 100.0°C? ans:
t = 16.8 sec
|
m = ρ V m = 1 g/cc (300cc) m = 300 grams |
Q =
m c DT Q = 300g (1) (100-20) Q = 24
kilocalories (4186J = 1 kCal) Q =
100,000 J |
P = Q
/ t (600W/m2)(10m2) = 100000J / t t = 16.8
sec |
Ch
20.3 #22
Two speeding lead
bullets, each of mass 5.00 g, and at temperature 20.0°C, collide head-on at
speeds of 500 m/s each. Assuming a
perfectly inelastic collision and no loss of energy by heat to the atmosphere,
describe the final state of the two-bullet system. Ans 805 °C
|
Must add the kinetic energy of the two bullets Kone = ½ m v2 Kone = ½ 0.005 (500)2 Kone = 625 Joules Ktwo = 1250 Joules cPb = 128 J/kg Lf-Pb = 2.45 x 104 J/kg |
Lead melts at
327 °C Qto melt point =
m c DT Q = (2*0.005)
(128) (327-20) Qto melt point = 393 Joules Energy to melt
lead Qfusion = m Lf Qfusion = 0.01 (2.45 x 104) Qfusion = 245 Joules |
Qremaining =
m c DT (1250-393-245)
= 0.01(128) DT DT = 478 °C So
final temp is 327 °C + 478 C° T =
805 °C |
Ch
20.4 #27
One mole of an
ideal gas is heated slowly so that it goes from the PV state (P0,
V0), to (3P0, 3V0), in such
a way that the pressure is directly proportional to the volume. (a) How much
work is done on the gas in the process? (b) How is the temperature of the gas
related to its volume during this process?
|
During the
heating process; P is proportional to V (given in the problem), so P = (constant) V where the constant
= Pi/Vi which yields P =
(Pi/Vi) V. Using same logic…if P is inversely prop to V then P = (Vi/Pi)
1/V, which is the normal situation (PiVi
= PfVf). |
|
|
(a) W = -∫
P dV W =
-∫(Pi/Vi) V dV from Vi to 3Vi W = -(Pi/Vi)
½ (Vf2 – Vi2) from Vi to 3Vi W = -½(Pi/Vi)
(9Vi2 – Vi2) W = -4 Pi Vi |
(b) P V = nRT (Pi/Vi)V V =
nRT T = (Pi / VinRT) V2 Temperature must be proportional to the
square of volume, rising to nine times its original value. |
|
Ch
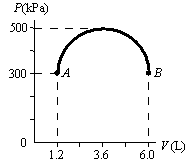
20.5 #33 A sample of an
ideal gas is in a vertical cylinder fitted with a piston. As 5.79 kJ of energy is transferred to the
gas by heat to raise its temperature, the weight on the piston is adjusted so
that the state of the gas changes from point A to point B along
the semicircle shown in Figure P20.33.
Find the change in internal energy of the gas. |
|
|
The work on the
gas, W = -∫P dV, which
is the area under the half circle curve plus the area below the half circle. Work
= -½ π(height/2)(width/2) + 300kPa (4.8 liters) Work = -½ π(200kPa)(4.8
liters/2) + 300kPa(4.8
liters) Work = -2194 kPa*liters Work =
-2194 Joules |
We know that the area of a circle is πr2. Area of a square is length * height. If we circumscribe a square about a circle of radius, r, then r
= height/2 or r = width/2So the area of a circle can also be written as π(height/2)(width/2) |
|
∆Eint = Q + Work ∆Eint = 5790 J + -2194 J ∆Eint = 3596 Joules |
Alternate
method
A
circle is % of a circumscribed square
π(h/2)2 / h x h
(h =
w in a square)
π/4 = 78.5 %
(78.5%
* 4.8 l * 200 kPa) + (300*4.8) = 2194 J
Ch
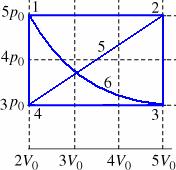
20.6 gasses
|
(a) Calculate the work done by the gas during
process 1 à 2 (b) Calculate the work done by the gas during
process 3 à 4 (c) Calculate the work done by the gas during
process 1 à 2 à 3 à 4 |
(d) Calculate
the work done by the gas during process 1 à 4 (e) Calculate the work done by the gas during
process 4 à 5 à 2 à 3 (f) Calculate the work done by the gas during
process 1 à 2 à 5 à 4 à 1 |
|
|
Calculate
the work done by the
gas during process 1 à 2 5P0
* (5-2)V0 = 15P0 V0 Calculate
the work done by the
gas during process 3 à 4 3P0
* (2-5)V0 = -9P0 V0 Calculate
the work done by the gas
during process 1 à 2 à 3 à 4 15P0
V0 + -9P0 V0 = 6P0
V0 |
Calculate
the work done by the
gas during process 1 à 4 5P0
* (0)V0 = 0 Calculate
the work done by the gas
during process 4 à 5 à 2 à 3 ½ b
h + width
h ½(5-2)V0
(5-3)P0 + (5-2)V0 3P0 12P0 V0 |
Calculate
the work done by the gas during process 1 à 2 à 5 à 4 à 1 15P0 V0 + 0 + -9P0
V0 + 0 = 6P0 V0 |
Ch
20.7 #51
The intensity of
solar radiation reaching the top of the Earth’s atmosphere is 1340 W/m2. The temperature of the Earth is affected by
the so-called greenhouse effect of the atmosphere. The result is that our planet’s emissivity
for visible light is higher than its emissivity for infrared light. For comparison, consider a spherical object
with no atmosphere, at the same distance from the Sun as the Earth. Assume its emissivity is the same for all
kinds of electromagnetic waves and that its temperature is uniform over its
surface. Identify the projected area
over which it absorbs sunlight and the surface area over which it
radiates. Compute its equilibrium
temperature. Chilly, isn’t it? Your calculation applies to:
![]() the
average temperature of the Moon
the
average temperature of the Moon
![]() astronauts
in mortal danger aboard the crippled Apollo 13 spacecraft
astronauts
in mortal danger aboard the crippled Apollo 13 spacecraft
![]() global
catastrophe on the Earth if widespread fires caused a layer of soot to
accumulate throughout the upper atmosphere, so that most of the radiation from
the Sun were absorbed there rather than at the surface below the atmosphere.
global
catastrophe on the Earth if widespread fires caused a layer of soot to
accumulate throughout the upper atmosphere, so that most of the radiation from
the Sun were absorbed there rather than at the surface below the atmosphere.
|
Our face showing
to the sun is a circle…πr2.
We’re radiating out throughout the entire surface area of the planet
4πr2. Stefan’s Law: P = σAeT4 ; σ = 5.669 x 10-8
W/m2K4 |
Pin = Pout σAineT4 = σ Aout
eT4 πr2e(1340) = σ(4πr2)eT4 (1340) = σ(4)T4 T =
277 K or 4 °C |