ball, coffee, 32, 33, 39, 43, 49
Ch
21.1 ball
(a) How many atoms
of helium gas fill an inflated basketball having a radius of 11.5 cm at 27.0°C
and 1.70 atm? (b) What is the average kinetic energy
of the helium atoms? (c) What is the root-mean-square speed of the helium
atoms?
|
(a) P
V = n
R T (1.013x105)1.7
4π(0.115)3/3 = n 8.314 300 K n = 0.440 moles N = 0.440 moles
* 6.022 x 1023 atoms/mole N = 2.65
x 1023 atoms (b) K = 3/2 kBT kB = R / NA K = 3/2 (8.314/6.022 x 1023)(300) K = 6.22 x 10-21 Joules |
(c) ½ mv2 = 3/2 kB
T vrms = Ö(3 R T
/ m NA) vrms = Ö(3*8.314*300/0.004 kg) vrms = 1370 m/s He = 2 atoms with 2 protons & 2 neutrons by
definition: 6.022 x 1023 amu’s = 1.000 g 4amu’s = 0.004 kg |
Ch
21.2 coffee
A 2-L Thermos
bottle is full of coffee at 95°C. You pour three cups
of coffee and then replace the cap. Make
an order-of-magnitude estimate of the change in temperature of the coffee
remaining in the Thermos bottle due to the air that replaces the poured coffee.
|
Assume Troom = 20 °C 1 liter ≈ ¼ gallon 1 gallon = 128 ounces 1 liter ≈ 32 ounces |
One cup is 6 ounces. 1/5 of 1000 ml = 200 ml = 200 cc. Three cups is 600cc. |
We’ll assume you fill your coffee cup completely so we’ll use 200
cc’s of air (replacing coffee) @ 20 °C and 1400 cc’s of coffee remaining @ 95
°C 1400 cc’s of coffee = 1.4 kg |
|
|
Energy
transferred from hot coffee
will be transferred to the
air that entered Thermos from room. |
|||
|
-m c ΔT = +(ρair Vair) CV air ΔTair
mair = ρVair -1.4 (4186) ΔT
= (.0012g/cc*600cc) 1.01 J/(gm K) (≈95 – 20) ΔT = -0.00931
°C so ΔT ≈ 10-2 °C |
N2 = 28 amu; O2 = 32 amu 80%N2 + 20%O2 = 28.8 amu/molecule 28 amu/molecule = 28.8 g/mole CV air = 7/2 (8.314J/mole K) (mole/28.8g) CV air = 1.01 J/(gram*K) |
||
|
Ch
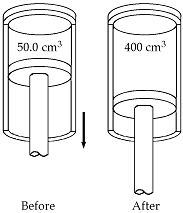
21.3 #32 During the power
stroke in a four-stroke automobile engine, the piston is forced down as the
mixture of combustion products and air undergoes an adiabatic expansion. Assume that (1) the engine is running at 2 500
cycles/min, (2) the gauge pressure right before the expansion is 20.0 atm, (3) the volumes of the mixture right before and
after the expansion are 50.0 and 400 cm3, respectively (4) the
time involved in the expansion is one-fourth that of the total cycle,
and (5) the mixture behaves like an ideal
gas with specific heat ratio 1.40.
Find the average power generated during the expansion. |
|
|
|
50 cc = 5x10-5
m3 20 atm (1.013x105 Pa / 1 atm) 20 atm ≈ 20x105 Pa 1 stroke for
a 4 stroke @ 2500 cycles/min ¼ (60 sec/1
min)(min / 2500cycles) 0.006 sec
per stroke |
Work = DEint = nCvDT Given:
Q = 0 J Work = n (5R/2)DT Work = 5/2 (nRDT) DPV = nRDT Work = 5/2
( Pf Vf –
P V ) Work = 5/2 (
1.14x105 40x10-5 – 20x105 5x10-5
) Work =
-150 Joules Power = Work /
time P = 150 J /
0.006 sec P =
25,000 Watts |
|
|
Assume air plus burnt gasoline behaves like a diatomic ideal gas.
PVγ = PfVγf
Pf =
P ( V / Vf
)γ Pf =
21atm (50/400)1.4 Pf
= 1.14 atm |
||
Ch
21.4 #33
Consider 2.00 mol
of an ideal diatomic gas. (a) Find the total heat capacity of the gas at
constant volume and at constant pressure assuming the molecules rotate but do
not vibrate. (b) What If? Repeat,
assuming the molecules both rotate and vibrate.
(a) (b)
|
N(5)kBT/2
= nCvT
nNA(5/2)kBT
= nCvT n(5/2)RT = n Cv T n Cv = n (5/2) R n Cv = 2 (5/2) 8.314 n Cv = 41.6 J/K
|
n CP
= n (Cv + R) n CP
= 2 (5/2 R + R) n CP
= 2 (7/2* 8.314) n CP
= 58.2 J/K |
In vibration with the center of mass fixed, both atoms are
always moving in opposite directions with equal speeds. Vibration adds two
more degrees of freedom for two more terms in the molecular energy, for
kinetic and for elastic potential energy. We have |
n Cv = n (7/2) R n Cv = 58.2 J/K
n CP
= n (9/2) R n CP
= 74.8 J/K |
Ch
21.5 #39
From the Maxwell-Boltzmann speed distribution, show that the most probable
speed of a gas molecule is given by Equation 21.29. Note that the most probable
speed corresponds to the point at which the slope of the speed distribution
curve dNv /dv is zero.
|
Maximum values
occur when the derivative is set to equal
zero (dNV / dV
= 0) |
NV = 4π N(m / 2πkBT)3/2 v2e-E/kBT NV = 4π N(m / 2πkBT)3/2 v2e-mv^2
/ 2kBT |
|
4πN (m/2πkBT)3/2
exp(-½mv2/kBT)
(2v – 2mv3/2kBT)
= 0 |
|
|
Solve for v to
find the most probable speed Reject as
solutions v = 0 and v = ∞ |
Retain only (2 – mv/kBT) = 0 Then vmp = (2kBT/m)1/2 |
Ch
21.6 #43
Assume that the
Earth’s atmosphere has a uniform temperature of 20°C and uniform composition, with an
effective molar mass of 28.9 g/mol. (a)
Show that the number density of molecules depends on height according to nv(γ) = n0 e-mgy / kBT where
n0 is the number density at sea level, where y = 0. This result is called the law of atmospheres. (b) Commercial jetliners typically cruise at
an altitude of 11.0 km. Find the ratio
of the atmospheric density there to the density at sea level.
|
(a) nV(E) = n0e- E /kBT n(h) = n0e-
mgh / kBT n(h)/n0
= e- mgh / kBT |
(b) n(h)/n0 = e-
Mgh / RT M = 0.0289
kg/mole, h = 11,000 m, T = 240 K n(h)/n0 = e-1.600 n(h)/n0
= 0.202 If students have 0.278,
ONLY give half credit for this problem, Instructor Solution mistake on temperature.
(anything else other than 0.278…like 0.269 or similar…give full credit) |
Ch
21.7 #49
Argon gas at
atmospheric pressure and 20.0°C is confined in a 1.00 m3 vessel. The
effective hard-sphere diameter of the argon atom is 3.10 ´ 10–10 m. (a) Determine the mean
free path ![]() . (b)
Find the pressure when
. (b)
Find the pressure when ![]() = 1.00 m.
(c) Find the pressure when
= 1.00 m.
(c) Find the pressure when ![]() = 3.10 ´ 10–10 m.
= 3.10 ´ 10–10 m.
|
Equation (from 21.7 notes) l = 1 / √2 πd2nV P = nVkBT nV = P / kBT l = 1 / √2 πd2 (P / kBT) l = kBT
/ √2 πd2 P |
(a) l = kB T
/ √2 π d2 P l = 1.38x10-23 293/ √2π(3.1x10-10)2
(1.013x105) l = 9.36 x 10-8
meters (b) P1 l1 = P2 l2 (All other
variables don’t change) 11 11
= P2 (9.36 x 10-8) P2
= 9.36 x 10-8 atm (c) P1 l1 = P2 l2 11 (3.1x10-10)1
= P2 (9.36 x 10-8) P2
= 302 atm |
Extra Problems…possibilities
for the final exam
A 2.00-mol sample
of a diatomic ideal gas expands slowly and adiabatically from a pressure of
5.00 atm and a volume of 12.0 L to a final volume of
30.0 L. (a) What is the final pressure of the gas? (b)
What are the initial and final temperatures?
(c) Find Q, W, and DEint.
|
(a) PVγ
= PfVγf Pf = P ( V / Vf
)γ Pf = 5atm (12/30)1.4 Pf = 1.39 atm |
(b) P = 5 atm
(1.013x105 Pa/atm) P Vγ
= n R T T = P
Vγ
/ n
R T = 5.065x105
(12x10-3)1.4 / 2mole(8.314) T =
363 K PfVγf
= n R Tf Tf = P
Vγ
/ n R Tf = 1.408x105 (30x10-3)1.4
/ 2 (8.314) Tf= 253 K |
|
(c) Given Q = 0 (adiabatic) ΔEint = Q + Work so γ = 1.40 and CV = 5R/2 ΔEint = n CV ΔT ΔEint = 2 5R/2 (253-365)K ΔEint = -4,660 Joules Given: Q = 0 J so Work =
-4,660 Joules |
The largest bottle
ever made by blowing glass has a volume of about 0.720 m3. Imagine that this bottle is filled with air
that behaves as an ideal diatomic gas.
The bottle is held with its opening at the bottom and rapidly submerged
into the ocean. No air escapes or mixes
with the water. No energy is exchanged
with the ocean by heat. (a) If the final
volume of the air is 0.240 m3, by what factor does the internal
energy of the air increase? (b) If the bottle is submerged so that the air
temperature doubles, how much volume is occupied by air?
|
(a) PVγ
= PfVfγ Pf =
P (V/Vf)γ Pf = P
(.72/.24)1.4 Pf
= 4.66P |
PV/T = PfVf/Tf Tf = Pf Vf
T / P V Tf = 4.66P(.24)T/P(.72) Tf = 1.55T |
Internal
energy, Eint = nCVT,
is directly proportional to temperature, so Eint,f / Eint =
Tf / T Eint,f / Eint = 1.55 |
|
(b) PVγ = PfVfγ
Pf/P
= (V/Vf)γ |
PV/T = PfVf/Tf Tf/T = (Pf/P) (Vf/V) Tf/T = (V/Vf)γ (V/Vf)-1 Tf/T = (V/Vf)γ-1 |
Tf/T = (V/Vf)γ-1 2 =
(.72/Vf)1.4-1 Vf = 0.127 m3 |
At what
temperature would the average speed of helium atoms equal (a) the escape speed from Earth, 1.12 ´ 104 m/s, and (b) the escape
speed from the Moon, 2.37 ´ 103
m/s? (Hint: Ch13: escape speed, mHe
= 6.64 ´ 10–27 kg.)
|
(a) vave2
= 8
kB T / π m 112002
= 8(1.38x10-23)T/π6.64 ´ 10–27 T =
23700 K |
(a) vave2
= 8
kB T / π m 23702
= 8(1.38x10-23)T/π6.64 ´ 10–27 T =
1060 K |