|
Thermodynamics Ch 17 – Heat and the 1st
Law of Thermodynamics |
|||||||||||
|
Heat and Internal Energy |
|||||||||||
|
Internal
energy is all the energy of a system that is associated with its microscopic
components – atoms and molecules – when viewed from a reference frame at rest
with respect to the center of mass of the system. Heat
is defined as the transfer of energy across the boundary of a system due to a
temperature difference between the system and its surrounds. |
|||||||||||
|
Units of heat |
|||||||||||
|
calorie |
the
amount of energy transfer required to raise the temp of 1 gram of water from
14.5 °C to 15.5 °C |
||||||||||
|
joule |
The
SI unit: 1 cal = 4.186 Joules 1 kcal = 1 Calorie = 4186 Joules |
||||||||||
|
BTU |
English
unit: (Q required to raise 1 lb, 1 °F) 1
Btu = 1054 Joules |
||||||||||
|
Specific Heat and
Calorimetric |
|||||||||||
|
Heat Capacity, C : Q = C
ΔT C,
the heat capacity is defined as the energy needed to raise the temperature of
a sample by 1°C |
Specific Heat: Q = m c
ΔT c,
the heat capacity of a sample per unit mass |
||||||||||
|
Question: Which would require more energy to bring to boil
if both are initially at room temperature? A) A cup of water B) A swimming pool full of
water Ans: I wouldn’t want to
have to pay the energy bill required to raise a swimming pool full of water
to its boiling point. Conclusion: The greater
the mass…the more energy required to bring to boiling point. |
Question: Which would require more energy to rise from 20
°C to 50 °C? A) A cup of water B) A cup of alcohol Ans: This one is harder…but
I believe many know raising the temperature of alcohol requires less heat than
water. Conclusion: The material
being heater makes a bit difference, so materials like water require much
more energy to change its temperature compared to materials like alcohol and
sand. |
||||||||||
|
Question: Which would require more energy to bring to
boil? A) A cup of water at 20 °C B) A cup of water at 90 °C Ans: This one is easy…the
one at 90 °C is almost at boil already, thus the energy input required to
bring it to boil will way less than the cup at 20°C Conclusion: The greater
the ΔT…the greater amount of energy was required. |
Question: Which would require more energy to bring to 20
°C? C) A cup of ice at 0 °C D) A cup of water at 0 °C Ans: This one is easy…imagine
adding 0°C water to your Coke or Pepsi to cool it down. 0°C ice requires much energy to bring it to
0°C water. Conclusion: The change of
state requires a large change of energy.
(This refers to section 12.8) |
||||||||||
|
Latent Heat |
|||||||||||
|
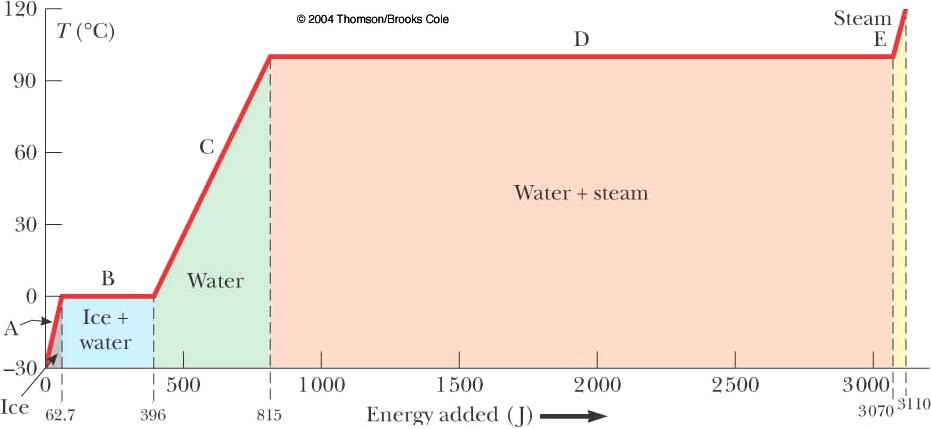
SI units are generally preferable, but I’m presenting this
section in cgs units Question: A large block of ice, 1000 g, is at -20 °C. How much energy is required to bring it to
100 °C steam? Ans: v à vaporization f à fusion units of c à calories / (gram C°) Q = cice m ΔT + m
Lf + cwaterm ΔT + m
Lv Q = ½ (1000)(20) + 1000(80) +
1 (1000)(100) + 1000 (539) Q = 10,000 cal + 80,000 cal + 100,000 cal +
539,000 cal Q =
729,000 calories (1 Calorie = 1 kilocalorie) Q =
729 Calories (4.186 J / Cal) Q = 3052 Joules (a good exercise for you is to work the
problem out using
SI units) |
|||||||||||
|
|
|||||||||||
|
Demo:
Latent Heat: TH-C-LH Demo:
Drinking Duck: TH-C-DD |
|||||||||||
|
Work and Heat in
Thermodynamics |
|||||||||||
|
Using
a macroscopic approach, the state of the system is described using P, V, T,
and internal energy, so these are called “state variables.” |
“Transfer
variables” only involve transfer of energy.
So no “change” or transfer of energy…no transfer variables. |
||||||||||
|
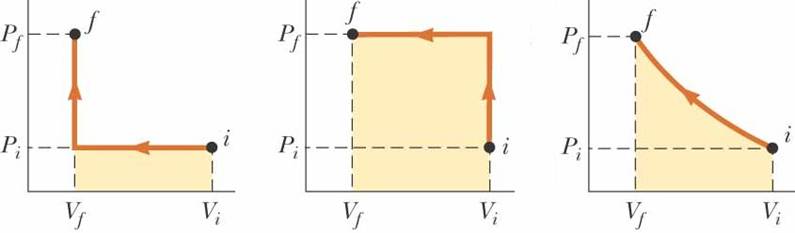
Work = force applied through a displacement Work = F ◦ dr Work = -F
dy Work = -PA dy Work = -∫ P dV from Vi to Vf |
The
work done on a gas as it is taken from an initial state to a final state
depends on the path between these states |
||||||||||
|
|
|||||||||||
|
|
|
||||||||||
|
Demo:
Diffusion in Air: TH-D-DA |
|||||||||||
|
The 1st Law
of Thermodynamics |
|||||||||||
|
The
1st law of thermodynamics (an energy conservation equation specifying that) the only type of energy that changes in the system are the internal
energy, Eint. |
ΔEint
= Q + W Internal
Energy is a state variable just like P, V, and T dEint
= dQ + dW |
||||||||||
|
thus Q + W is INDEPENDENT of path |
|||||||||||
|
An
isolated system is a system that no energy is
transferred by heat and the work done on the system is zero. (Doesn’t interact with environment) Q
= W = ΔEint = 0 Joules ΔEint-i
= ΔEint-f |
Non-isolated,
but is cyclic (starts and ends in the same state) ΔEint
= 0 Joules Thus
Q (energy added to system) is equal
to the negative Work. Q
= -Work |
||||||||||
|
Some Apps of the 1st
Law of Thermodynamics |
|||||||||||
|
ΔEint =
Q + W |
|||||||||||
|
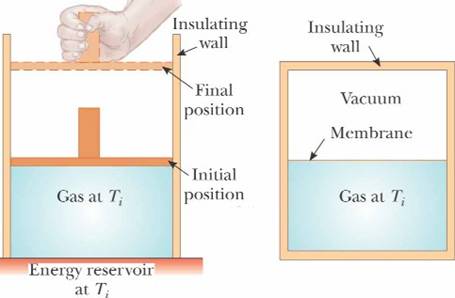
An
adiabatic process is one that no energy enters or leaves from the system by
heat. Q
= 0 Joules |
ΔEint
= Work As
gas expands adiabatically the temperature reduces accordingly |
For a cloud in deep space that is spurting out material very fast,
at that point the temperature can go below the ambient background radiation
temperature of 3 Kelvin |
|||||||||
|
Isobaric
(Constant Pressure) W
= -P ΔV P
is a constant |
A plot of P vs V for an ideal gas yields a hyperbolic curve (an
isotherm) Isothermic (constant
Temp) ΔEint = 0 Joules Q = -W W = -∫ P
dV W = -∫ (nRT/V) dV Work = -nRT ln(Vf/Vi) Work = nRT ln(Vi/Vf) |
Isovolumetric ΔEint
= Q If energy is added by heat to a system, all of the transferred
energy remains in the system as increased internal energy |
|||||||||
|
Energy-Transfer
Mechanics |
|||||||||||
|
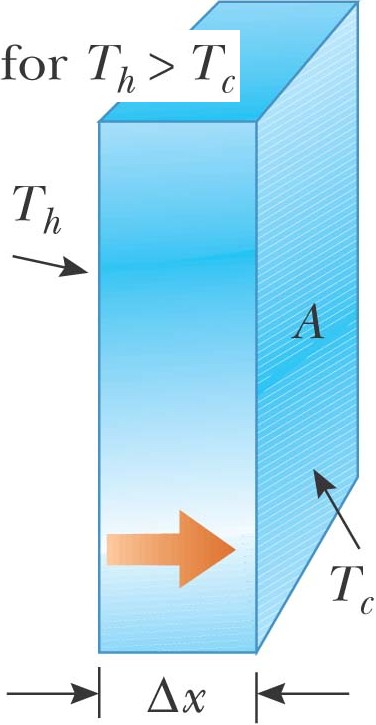
Conduction P
= Q / t = kA dT/dx dx:
The thicker the wall…more isolated A: More area…the more energy flows through the
wall dT: The greater the temperature difference …the
more energy flow through the wall k: And different materials will have different
coefficients for heat flow P
= kA ΔT/Δx P
= A ΔT/(Δx/k) P
= A ΔT/(ΣRi) R
à ft2 °F hr/Btu 1 ft2 °F
hr/Btu (0.305m/ft)2 (5°C/9°F)(3600s/hr)(1Btu/1054J)
= 0.1765 m2 °F sec / J R-11
english = R-2 in metric |
|
||||||||||
|
Convection Energy
transferred by the movement of
a warm medium Demo:Convection
Currents in Air: TH-B-CC |
|||||||||||
|
Radiation Stefan’s
Law: P = σAeT4 σ
= 5.669 x 10-8 W/m2K4, e
is emissivity (0 to 1) Ideal
absorber, e = 1 (Black Body) Ideal
reflector, e = 0 |
|||||||||||