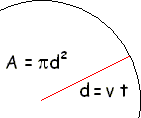|
Thermodynamics
Ch 18 – The Kinetic
Theory of Gases |
||||||||||||||||||||
|
Molecular Model of an
Ideal Gas |
||||||||||||||||||||
|
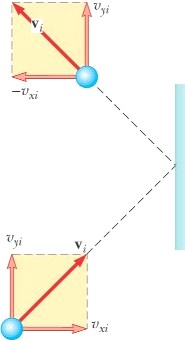
Let’s
look at a molecule striking a wall of a container. The a particle of mass, m, and a velocity, vx, strikes a wall elastically we know that
the impulse (F Dt) (F Dt) is Dp = (-mvx – mvx)
= -2mvx (it also could be +2mvx) F Dt = -2mvx
F (2x/vx) = -2mvx
F = -mvx2/x According
to Newton’s 3rd law, the wall will exert an equal and opposite
force on the particle. Fwall
= mvx2/x Fwall
= (m/x) vx2 by one particle And
if all same type of particle (homogeneous) the average m/x for each particle will
be equal, and the average velocity of each particle will be
|
|
|||||||||||||||||||
|
So
the total force by all the particles is F
= N (m/x) v2 = vx2
+ vy2 + vz2 ; since completely random v2 = 3vx2 F
= N /3 (m and since P = F / A |
Which means temperature is a direct measure of the average
molecular kinetic energy. And
since ½kBT
= ½m ½kBT
= ½m ½kBT
= ½m The theorem of equipartition
energy states that each degree of freedom (x, y, z) contributes ½kbT
to the energy of the system Etranslational = N (½m Etranslational = 3N(½kBT) Etranslational = 3/2 NkBT Etranslational = 3/2 nRT vrms = Ö vrms = Ö(3kBT/m) 3/2 kBT = ½m vrms = Ö(3RT/M) M = mNA Remember kB = R / NA kB = 8.314 / 6.022 x 1023 kB = 1.38 x 10-23 J/K |
|||||||||||||||||||
|
P
= F / A P
= F / d2 P
= N/3 (m Eq 21.2 in book P = 2/3 N/V (½m vave2) |
PV = nRT kB = R/NA PV = NkBT N = n NA |
|||||||||||||||||||
|
PV = PV 2/3 N (½m T = 2/3kB (½m 3/2 kBT
= ½m |
||||||||||||||||||||
|
Example:
A spherical balloon of volume 4000 cm3 contains helium
at an (inside) pressure of 1.20 ´ 105
Pa. How many moles of helium are in the balloon if the average kinetic energy
of the helium atoms is 3.60 ´ 10–22
J? P = 2/3 N/V (½mvave2) Eq 21.2 in book (derived
above) N
= 3/2 P V / KE N
= 3/2 (1.2x105)(0.004) / 3.6x10-22 N
= 2 x 1024 molecules n = N/NA = 2 x 1024/ 6.02 x 1023 n = 3.32 moles |
||||||||||||||||||||
|
Demo: Crooke’s Radiometer: TH-D-CR |
||||||||||||||||||||
|
Molar Specific Heat of
an Ideal Gas |
||||||||||||||||||||
|
Q
= n CVΔT The
energy added to the gas by heat at a constant
volume only the internal energy, Eint,
of the gas increases. (No
work is done; Area under curve is zero since a vertical line.) |
|
|||||||||||||||||||
|
W
= -∫PdV Work
= 0 J ΔEint = n CVΔT
+ W ΔEint = Q + W (but W = 0) ΔEint = Q |
Constant
Volume We
know Eint = Ktrans
= 3/2 nRT Eint = n CVT 3/2
nRT = n CVT CV = 3/2 R |
|||||||||||||||||||
|
This
result is if the only internal energy is translation …thus monatomic
atoms. Thus we predict CV =
3/2 R for all monatomic atoms, CV = 3/2 (8.314 J/ K*mole) CV = 12.5 J/ K*mole We find that this is true
experimentally (Table 12.2) |
||||||||||||||||||||
|
Q
= n CPΔT The
energy added to the gas by heat for constant
pressure not only increases the internal energy, Eint,
but also must account for the energy transferred out of the system by the
increase volume, Work (area under the curve.) We
see that T+ΔT is the same for both processes (constant pressure and
constant volume), but the constant pressure path has a change of volume which
represents the negative amount of work that is also done on the system such
that |
||||||||||||||||||||
|
ΔEint = Q +
W ΔEint = nCPΔT + -P ΔV n
CVT = nCPΔT +
-nRΔT CP
- CV = R CP
– 3R/2 = R CP = 5R/2 |
We know ΔEint for both constant pressure and
constant volume is the same, n CVΔT, due to both go to the
same isotherm. |
A
ratio of these two specific heats is useful in solving problems, thus γ = CP/CV = 5R/2 /
3R/2 γ = 1.67 |
||||||||||||||||||
|
Once
again, the result is seen experimentally to be true. |
||||||||||||||||||||
|
Example: Calculate the change in internal energy of 3.00 mol of
helium gas when its temperature is increased by 2.00 K. |
Eint = 3/2 n
R ΔT Eint = 3/2 (3)(8.314)(2K) Eint = 74.8 Joules |
|||||||||||||||||||
|
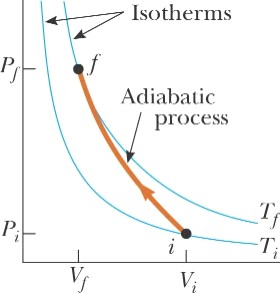
Adiabatic Processes for
an Ideal Gas |
||||||||||||||||||||
|
Adiabatic
Process is a process where no energy is transferred to surrounding
environment. These processes are
either quick processes or insulated processes and is represented by PVγ
= constant |
||||||||||||||||||||
|
ΔEint
= Q +
W ΔEint
= 0 + nCVΔT -PdV = nCV
dT |
but
for adiabatic processes
(insulated
where Q = 0), the
internal energy only depends on Temperature |
|||||||||||||||||||
|
PdV + VdP = n R
dT |
The
total differentiation of the ideal gas state, PV = nRT
is |
|||||||||||||||||||
|
PdV + VdP =
nR(-PdV/nCV) PdV + VdP =
-(R/CV) P dV |
Solve
-PdV = nCV dT for dT = -PdV/nCV
and substitute in |
|||||||||||||||||||
|
dV/V + dP/P = (CV – CP)/CV dV/V dV/V + dP/P =
(1 – γ) dV/V γ ∫dV/V + ∫dP/P = 0 |
R = CP – CV
and divide by PV γ = CP/CV |
|||||||||||||||||||
|
γ ln (V) + ln (P) = 0 which is equivalent to PVγ
= constant |
||||||||||||||||||||
|
Example Air in a thundercloud expands as it rises. If its initial temperature is 300 K, and
no energy is lost by thermal conduction on expansion, what is its temperature
when the initial volume has doubled? |
PVγ
= constant PVγ
= PfVfγ
T Vγ-1 = Tf
Vfγ-1 300 Vγ-1 =
Tf (2V)γ-1 (V/2V)1.40-1 = Tf /300 Must
use γ for diatomic molecules,
1.40 Tf = 227 °C |
|
eq. 21.20 P Vγ = Pf Vfγ
(nRT/V)Vγ = (nRTf/Vf)Vfγ (T/V) Vγ =
(Tf/Vf) Vfγ
T Vγ-1 = Tf Vfγ-1 |
|||||||||||||||||
|
The Equipartition
of Energy |
||||||||||||||||||||
|
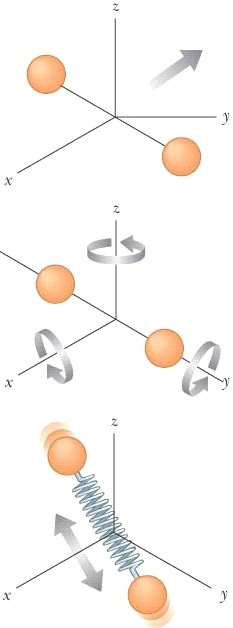
As
we’ve seen the model for molar specific heat agrees only with monatomic gases
as expected. Also
our value of CP – CV = R does prove to be accurate for
all gases. We
need to include (a)
translational KE (b)
rotational (c)
vibrational. |
|
As
we can see the distance the diatomic atom is from the y-axis so the Inertial,
KR
= ½Iyω2 = 0 Joules, but
Inertia is far from zero for rotation about the x & z-axes Thus
the total inertia for any diatomic molecule will just be two of the three
axes. (We
can rotate the axis so that Inertia will always be zero about one of the
axes.) |
|
|||||||||||||||||
|
To
write Eint with both (a) translational
and (b) rotational energy yields Eint = ETrans
+ ERot (each degree of freedom
contributes Eint = 3N(½kbT) + 2N(½kbT) on average ½kbT per molecule, for an Eint = 5N(½kbT) additional
two degrees of freedom Eint = 5/2 nRT for rotation) |
||||||||||||||||||||
|
CV
= 1/n (dEint/dT) CV
= 5/2 R CP
– CV = R CP
– 5/2 R = R CP
= 7/2 R γ = CP/CV
= 7R/2 / 5R/2 γ = 1.40 |
We
know from the previous section monatomic atoms γmono = 1.67 And
for diatomic atoms γdi = 1.40 The
results agree well with experimental data. |
|||||||||||||||||||
|
To
write Eint with both (a) translational
and (b) rotational energy (c) and vibrational
yields Eint = ETrans
+ ERot + EVib (2 more degrees of freedom for Eint = 3N(½kBT) + 2N(½kBT) + 2N(½kBT) vibration; each atom is vibrating) Eint = 7/2 nRT Eint = 7N(½kBT) |
||||||||||||||||||||
|
CV
= 1/n (dEint/dT) = 7/2 R CP
– CV = R CP
– 7/2 R = R CP
= 9/2 R γ = CP / CV γ = 9R/2 /
7R/2 γ = 1.29 |
We
know from the previous section monatomic
atoms, γmono = 1.67 diatomic
atoms, γdi = 1.40 And
for polyatomic atoms, γpoly = 1.29 The
results agree well with experimental data. |
|||||||||||||||||||
|
To
explain molar specific heat of a solid at high temps, we use small
displacements of an atom from its equilibrium position which is approximated
by simple harmonic motion. Ex
= ½mvx2 + ½kx2 (review from Ch 7 & 8) Similarly
along the y & z-axes, thus for a total of 6 degrees of freedom. Eint = Ex + Ey +
Ez (Remember…energy isn’t a vector) Eint = 2N(½kBT) + 2N(½kBT) + 2N(½kBT) Eint = 3NkBT = 3nRT CV
= 1/n (dEint/dT) CV
= 3 R (at lower temperatures classical physics just isn’t
a good enough approximation; this is only good at high temps) |
||||||||||||||||||||
|
Example A certain molecule has f degrees of freedom. Show that an ideal gas consisting of such
molecules has the following properties:
|
||||||||||||||||||||
|
(1)
total internal energy is fnRT/2; Eint = N(½kBT) for each degree of freedom theorem of equipartition energy So if,
f, molecules…then Eint = fN(½kBT);
where n = N/kB From Ch 19: kB = R
/ NA ; n = N / NA; N = nR / kB Eint = fn(½RT) (2)
molar specific heat at constant vol. is fR/2; CV
= 1/n ( dEint / dT) CV
= 1/n (d
[fn(½RT)] /
dT) CV = ½fR |
(3)
molar specific heat at constant pressure; is (f + 2)R/2;
CP
– CV = R CP
= ½fR + R CP
= fR/2 + 2R/2 CP = ½(f + 2)R (4)
specific heat ratio is g = CP/CV
= (f + 2)/f γ = CP /
CV γ = ½(f + 2)R / ½fR γ = (fR
+ 2R) / fR γ = (f + 2) / f |
|||||||||||||||||||
|
The Boltzmann
Distribution Law |
||||||||||||||||||||
|
The
number density from statistical mechanics is nV(E)
= n0e-E/kBT Thermal
Excitation of Atomic Levels, p. 655 Demo: Bromine Diffusion: TH-D-BD |
||||||||||||||||||||
|
Distribution of
Molecular Speeds |
||||||||||||||||||||
|
To
the right is the observed speed distribution of gas molecules in thermal
equilibrium. NV is called
the Maxwell-Boltzmann speed distribution. The
number of molecules having speeds in the range of v to v + dv is equal to the area of the shaded rectangle, NV
dv. As
NV approaches zero as v approaches infinity (to the right à ). |
|
|||||||||||||||||||
|
NV = 4π N(m /
2πkBT)3/2 v2 e-KE/kBT
NV = 4π N(m /
2πkBT)3/2 v2 e-½mv^2 / kBT
This
is the curve in the active figures |
Example: From the Maxwell-Boltzmann speed
distribution, show that the most probable speed of a gas molecule is given vmp = (2kBT/m)1/2.
Note that
the most probable speed corresponds to the point at which the slope of the
speed distribution curve dNv /dv
is zero. |
|||||||||||||||||||
|
vrms = (3kBT / m)1/2 vrms = 1.73 (kBT / m)1/2 vave = (8kBT /
π m)1/2 vave = 1.60 (kBT / m)1/2 vmp = (2kBT / m)1/2
vmp = 1.41 (kBT / m)1/2 vrms > vave > vmp |
Solution In
the Maxwell Boltzmann speed distribution function
take dNV / dv
= 0 to find 4πN (m/2πkBT)3/2 exp(-½mv2/kBT) (2v – 2mv3/2kBT) = 0 exp(-½mv2/kBT) (2v – 2mv3/2kBT) = 0 2exp(-mv/2kBT) 2v
(1 – mv2/2kBT) =
0 exp(-mv/2kBT) v
(1 – mv2/2kBT) =
0 and solve for v to find the most
probable speed Reject as solutions v
= 0 and v = ∞ Retain only (1
– mv2/2kBT) =
0 Then vmp = (2kBT/m)1/2 |
|||||||||||||||||||
|
Demo:
Diffusion in Air: TH-D-DA |
||||||||||||||||||||
|
Mean Free Path |
||||||||||||||||||||
|
The
mean free path, L, is the average distance, d = vavet,
divided by the number of collisions that occur in that time interval |
|
L
= vavt / (Acircle
d)nV If motion of L
= vavt / (πd2 vavt)nV molecules are included L = 1 / (
πd2 )nV L = 1 / √2 πd2nV |
||||||||||||||||||
|
With
a collision frequency of |
f = πd2vavenV |
f
= √2 πd2vavenV f
= vave (√2 πd2nV) f
= vave / L |
||||||||||||||||||
|
Where
the inverse of the frequency is the mean free time (period) |
Tmeanfree = 1 / πd2vavenV |
|||||||||||||||||||
|
In an ultrahigh vacuum system, the pressure is measured to be 133
´ 10–10 Pa. Assuming the
molecular diameter is 3.00 ´ 10–10
m, the average molecular speed is 500 m/s, and the temperature is 300 K, find
the number of molecules in a volume of 1.00 m3 and the mean
free path of the molecules |
N = P V NA / R T N = 133x10–10(1)6.02x10–23/8.314(300) N = 3.21 x 1012 L = 1
/ √2 πd2nV L = V / N √2 πd2 L = 1 / 3.21x1012 √2 π (3.00x10–10)2 L = 7.79 x 105 meters |
PV = n R T PV = N/NA RT |
||||||||||||||||||