|
Nuclear Power
What are some common household items that have radioactive parts…. What about older smoke
detectors…Americium 241, an alpha emitter. Also anything with potassium
like bananas, or potatoes….because the natural K,
includes 0.012% K-40. |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Example 1 How many neutrons are
ejected from the U-238 nucleus on disintegration? |
Solution; only focus on neutrons 146 à 85 + 55 + x 6 neutrons are ejected
Note: if a beta particle is also ejected, does that effect neutrons?
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
· Power generation o U-235 is about 5 to 6.5% § (so U-238 is about 93 to 95%) o Yellow cake is about 99.3% U-238, so U-235 must be enriched.
The enrichment process involves uranium hexafluoride gas and centrifuges.
How are U-235 and U-238 separated in a centrifuge?
· If enrichment is beyond 8 or 9%, only one purpose. Weapons. o Weapons grade starts ~ 40% of U-235
|
USA à PWR and BWR, outside of US, many breeder reactors
USA · we don’t re-process · Outside of US, reprocess the spent fuel rods.
In reactors: · U-235 has roughly ½ of initial remaining after “spent” (Starts at ~5% U-235, removed at ~2.5%) · Most reactors are research and for diagnostics both industry and hospitals.
What do we notice about the mass of the neutron? Also what is the charge of a neutron?
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
AMUs |
(x 10-30) kg |
|
Referring to our
homework, E = mc2 What is mass difference
between?
mp + me +
E ≈ mn (1672.6 + .91 + E ≈ 1674.9)x10-30 E ≈ 1.4 x 10-30 c2 E = 1.4 x 10-30 (3x108)2 = 1.26 x 10-13 Joules
1.26 x 10-13 Joules (1 eV / (1.602e-19 J) Or 7.9 x 105 eV or 0.79 MeV
|
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Neutron Proton (+) Electron (-) |
1.00866 1.00728 0.00055 |
1674.9 1672.6 0.91 |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
What is an eV (electron Volt)?
The energy an electron gains being accelerated through a potential difference of 1 Volt through 1 meter |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
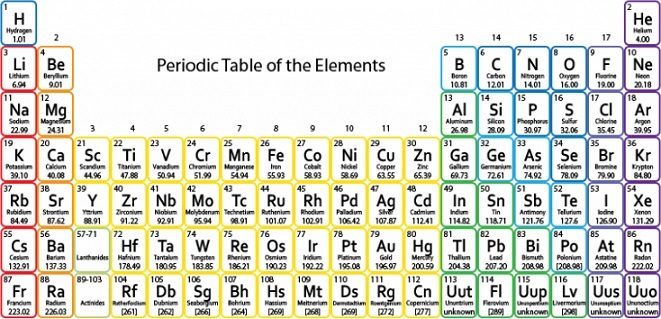
https://sciencenotes.org/printable-periodic-table/ Free Printable Periodic Tables (PDF and PNG)
|
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
An element is defined by # of protons. As element become more massive, for geometric reasons (spacing the positive protons further apart) and the strong nuclear force (hold the neutrons together), more neutrons are required to be stable.
The four forces · Gravity (F = G M m / r2) · Weak (nuclear) · Electro-magnetic (F = k Q q / r2) · Strong (nuclear)
|
|
What if….
Question 1 (92 p+, 140 n) U-242 undergoes beta decay, what is it?
Question 2 (93 p+, 139 n) Np-242 undergoes a beta decay, what is it?
Question 3 (94 p+, 138 n) Pu-242 undergoes an alpha decay, what is it?
Question 4 (92 p+, 136 n) A proton in U-238 absorbs a high energy cosmic ray (gamma), what does it emit and what is it? Hint: undergoes positron emission
|
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Look at U-235 (0.72%)
and U-238 (99.27%; T½ = 4.5 billion year (half-life)) which we discussed the
other day. We discussed forming uranium hexafluoride, a
gaseous form, with U-238 being 3 AMUs more massive than U-235 and centrifuges (also improperly named, remember chalk board pushing you without you initially pushing the board) Any uranium
dust over 7-8% U-235, only
purpose is for weapons grade uranium…
|
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Question 5 (91 p+, 137 n) Pa-238 undergoes fission and emits one neutron, and one alpha and one beta; if one fission fragment is Tc-103 (43p+, 60n), what is the other fragment? During beta emission a neutron decays into a proton and electron (with neutrino). So the right side of the equation loses a neutron, and gains a proton, so 92 protons, and 146 neutrons
Easiest way to do this…evaluate the the beta particle FIRST, then do the new problem. So how to get a β à neutron decays to p+ and β ; Thus Pa-238 à U-238
After (due to positron emission)----CHECK this…is this correct?????????? Due to conservation: Left side = right side or Left side - right side = 0 Protons: 92 – 43 – p+ -2 = 0 à p+ = 47 Neutrons: 146 – 60 – n – 2 – 1 = 0 à n = 83 |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Question 6 (92 p+, 136 n) A proton in Magnesium-23 absorbs a high energy cosmic ray (gamma), what does it emit and what is it? Hint: undergoes positron emission
Note: the emitted neutrino, ve, is neglected |
|
Question 7 If high pressure and temperature fuse two hydrogen atoms together, what is this called?..... this is rhetorical, the answer is in the question, Fusion.
|
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
|
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||