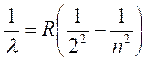Pre-Lab
- Atomic Spectra Name
|
|
||||||||
|
|||||||||
|
|
Balmer (ending on 2nd
energy state) |
||||||||
|
Color |
n |
λ (nm) |
|||||||
|
red |
3 |
656.2 |
|||||||
|
“R” is the Rydberg
Constant |
|
4 |
|
||||||
|
|
|
5 |
|
||||||
|
violet |
6 |
|
|||||||
|
limit |
∞ |
|
|||||||
|
(#6 & 7 are double value questions) m λ = “d” sinθ d: inverse of the grating constant in meters (#5) θ:
degrees from central axis passing through the scope |
|||||||||
|
Please
fill in the following table using the above formula and value of “d”. |
|||||||||
|
|
λ (nm) (from #6) |
θ for m = 1 |
θ for m = 2 |
Show work for 1st order violet (m = 1) |
|||||
|
Red |
|
|
|
||||||
|
Green |
|
|
|
||||||
|
Blue |
|
|
|
||||||
|
Violet |
|
|
|
||||||
|
1/λ = -R 1/ni2 + R/4 y
=
m x + b |
(answer must be in the
form of expressions ONLY) |
||||||||